For US Healthcare Professionals Only
Safety & Tolerability
Straightforward Treatment in Patients
With Complications
BAXDELA™ is well tolerated
The most common side effects were nausea and diarrhea and were generally mild/moderate, of limited duration, and did not lead to discontinuation of drug
Most Common Treatment-Emergent Adverse Events (TEAEs) Occurred in ≥2.0% (pooled data from phase 3 trials)
| BAXDELA N=741 |
Vancomycin + aztreonam N=751 |
|
|---|---|---|
| Nausea | 8% | 6% |
| Diarrhea | 8% | 3% |
| Headachea | 3% | 6% |
| Transaminase elevationsa | 3% | 4% |
| Vomiting | 2% | 2% |
|
aThe data are not an adequate basis for comparison of rates between the study drug and the active control. bPooled reports include hypertransaminasaemia, increased transaminases, and increased ALT and AST. |
||
Discontinuations Due to Treatment-Related Adverse Events (n/N)
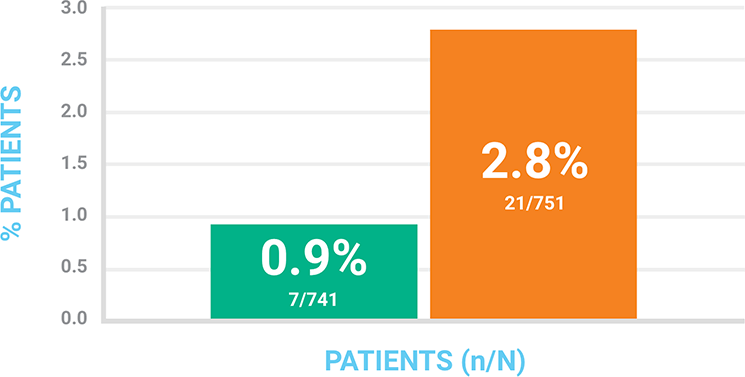
Discontinuation rate is not statistically significant.
Important Additional Safety Information
QT prolongation was not observed in patients taking BAXDELA in a definitive QT study
- Some fluoroquinolones have been associated with prolongation of the QT interval on electrocardiogram and infrequent cases of arrhythmia
- A thorough QT study found no QT prolongation in patients taking BAXDELA compared with moxifloxacin
Photosensitivity was not observed in patients taking BAXDELA in a photosafety study
- Fluoroquinolones have been associated with the potential for phototoxicity; however, a phase 1 study conducted in 52 healthy volunteers demonstrated no clinically relevant photosensitizing potential for BAXDELA compared with lomefloxacin
- Potential for phototoxic reactions should always be considered when caring for patients taking BAXDELA or any other marketed fluoroquinolone
Hepatotoxicity
- Postmarketing reports of severe hepatotoxicity (including acute hepatitis and fatal events) have been received for patients treated with fluoroquinolones
- No cases of Hy’s lawa were reported in the BAXDELA clinical trials
Dysglycemia
- Dysglycemia, including hypoglycemia, has been associated with all fluoroquinolones, which are believed to stimulate insulin release via interference with the K+/ATPase pump in pancreatic β cells
- In phase 3 trials, the incidence of dysglycemia was similar to the incidence observed with vancomycin plus aztreonam
aHy's law is a rule of thumb that a patient is at high risk of a fatal drug-induced liver injury if given a medication that causes hepatocellular injury with jaundice.
Melinta Therapeutics, Inc. - The Antibiotics Company
For additional information about BAXDELA, call Melinta Therapeutics at 1-844-MELINTA (1-844-635-4682) or contact the US Food and Drug Administration at 1-888-INFO-FDA (1-888-463-6332).
©2017 All rights reserved PP-BAX-US-0072 v1.1 Terms of Use
