BAXDELA 300 mg IV q12h vs comparator
For US Healthcare Professionals Only
Assessment at Baseline: Lesion size ≥75 cm2

TRIAL 1: IV Only
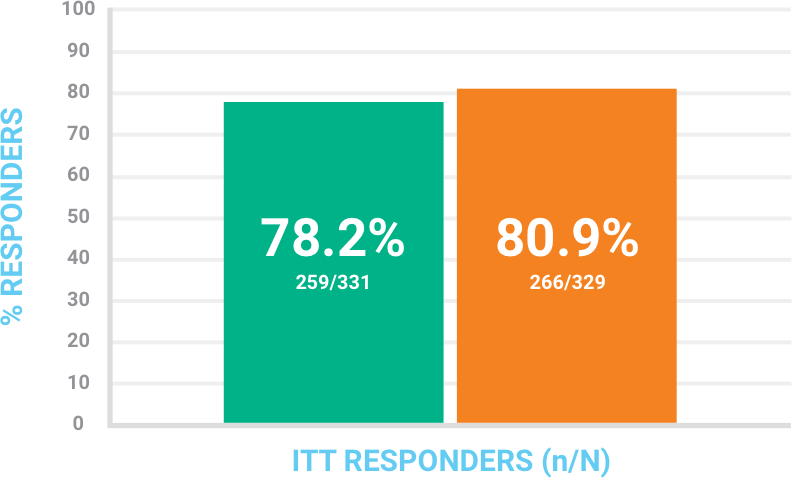
BAXDELA IV
(n=331)
TRIAL 2: IV to Oral
BAXDELA IV to oral
(n=423)
BAXDELA 300 mg IV q12h
BAXDELA 300 mg IV q12h for 6 doses, then switched to 450 mg oral q12h

Vancomycin plus aztreonam
(n=329)
vancomycin
15 mg/kg plus aztreonam

Vancomycin plus aztreonam
(n=427)
vancomycin
15 mg/kg plus aztreonam

PRIMARY Endpoint: At 48-72 h, reduction in lesion size ≥20%

SECONDARY Endpoint: At day 14 (+/- 1) follow-up, investigator-assessed clinical success defined as complete or near-complete resolution of signs and symptoms with no further antibacterial therapy needed



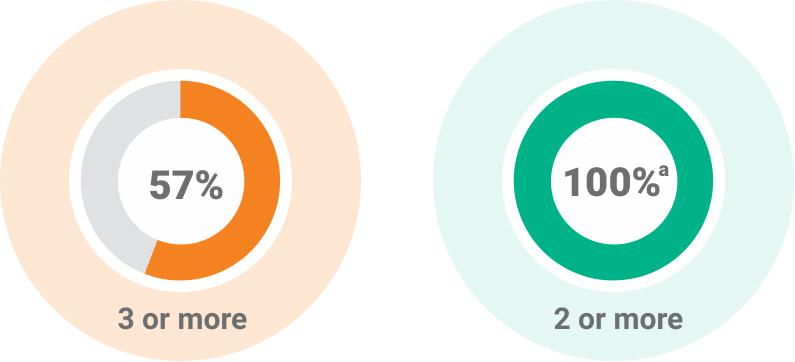
aWith the exception of one patient, all patients randomly assigned to treatment met the requirement for having at least 2 systemic signs of infection.
Objective responsea at 48-72 hours in the ITT population
At least 20% reduction in lesion size.a
BAXDELA 300 mg IV q12h vs comparator

BAXDELA 300 mg IV q12h for 6 doses, then a mandatory switch to oral BAXDELA 450 mg q12h vs comparator
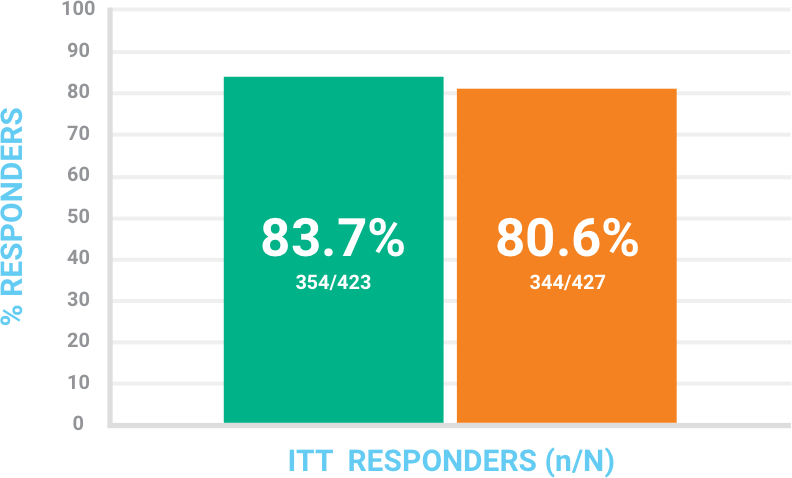
CI = confidence interval; ITT = intention-to-treat.
aDetermined by digital planimetry of the leading edge of erythema without other reasons for failure (use of another antibiotic or surgical procedure to treat for lack of efficacy). Missing patients were treated as failures in the ITT analysis set.
Investigator assessment of response at Follow-Up Visit (day 14 +/− 1) in the CE population
Success was defined as cure + improved where patients had complete or near-complete resolution of signs and symptoms, with no further antibiotic therapy needed.
BAXDELA 300 mg IV q12h vs comparator
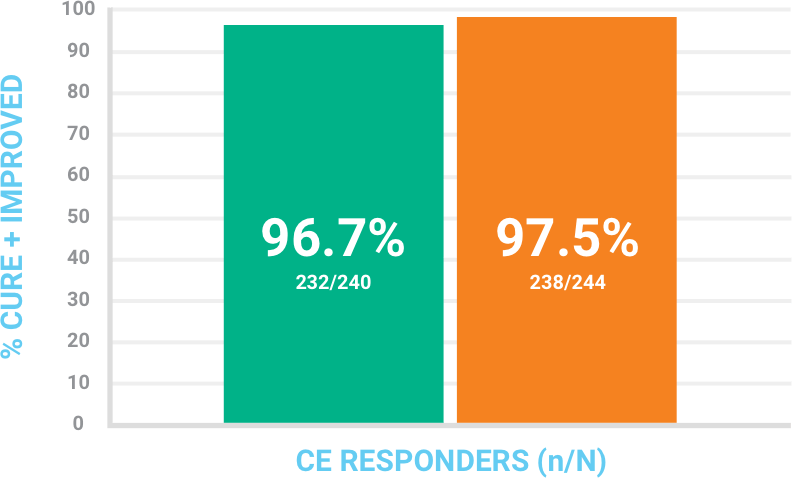
BAXDELA 300 mg IV q12h for 6 doses, then a mandatory switch to oral BAXDELA 450 mg q12h vs comparator
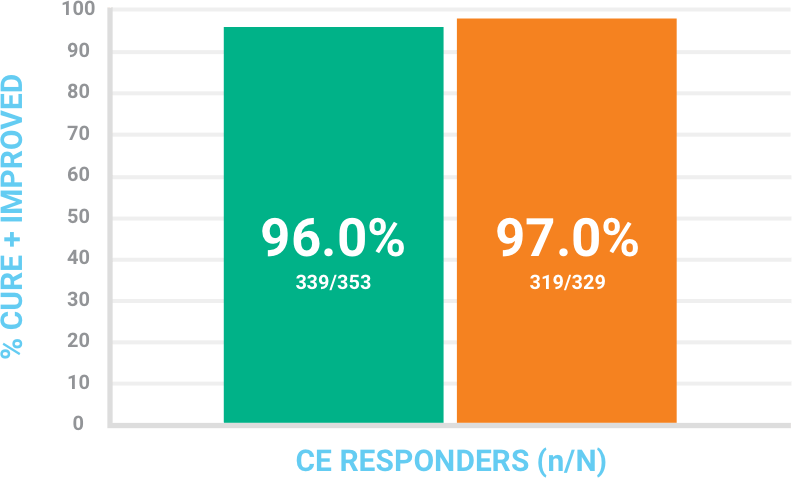
CI = confidence interval; CE = clinically evaluable consisted of all ITT patients who had a diagnosis of ABSSSI, received at least 80% of expected doses of the study drug, did not have any protocol deviations that would affect the assessment of efficacy, and had investigator assessment at the Follow-Up Visit.
BAXDELA monotherapy was comparable to vancomycin plus aztreonam in the eradication of MRSA infection
Analysis set consisted of patients who were Microbiologically Evaluable at Follow-Up visit for Investigator-assessed response (MEFUI)
POOLED DATA TRIALS 1 AND 2
Microbiological Response at Follow‑up in Patients With MRSA Infections
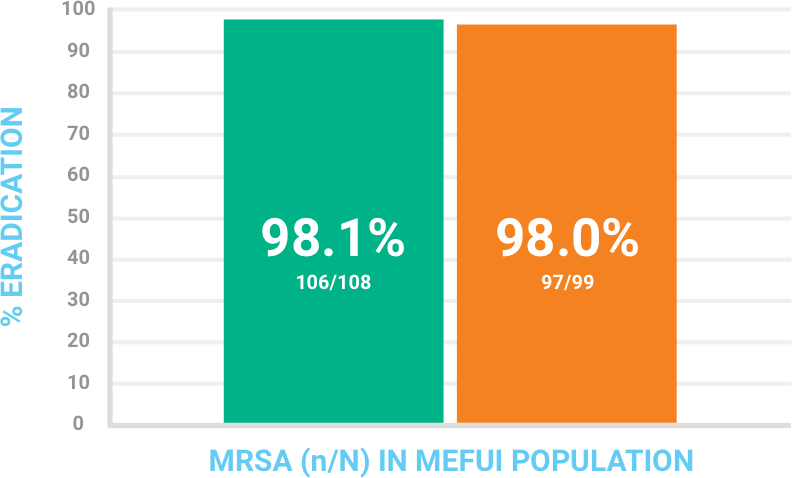
Data on file. Melinta Therapeutics, 2016.
In the Microbiological ITT (MITT) population, microbiological response was as follows: BAXDELA, 122/144 (84.7%); vancomycin plus aztreonam, 116/141 (82.3%)
Microbiological response for all groups was defined as eradicated or persisted. Each group included both documented and presumed results.
Analysis set: Microbiological ITT (MITT) consisted of all randomized patients who had a baseline pathogen identified that is known to cause ABSSSI
Success was defined as complete or near-complete resolution of signs and symptoms with no further antibacterial therapy needed at follow-up visit.
| Staphylococcus | BAXDELA n/N |
Comparator n/N |
|---|---|---|
| S aureus | 275/319 (86.2%) | 269/324 (83.0%) |
| methicillin-susceptible (MSSA)a | 154/177 (87.0%) | 153/183 (83.6%) |
| methicillin-resistant (MRSA)a | 122/144 (84.7%) | 116/141 (82.3%) |
| S lugdunensis | 10/11 (90.9%) | 8/9 (88.9%) |
| S haemolyticus | 13/15 (86.7%) | 7/8 (87.5%) |
| aDiscrepancy in the total numbers is due to multiple subjects having both MRSA and MSSA isolates. | ||
| Enterococcus and Streptococcus Pathogens | BAXDELA n/N |
Comparator n/N |
|---|---|---|
| E faecalis | 9/11 (81.8%) | 14/16 (87.5%) |
| S agalactiae | 12/14 (85.7%) | 11/12 (91.7%) |
| S anginosus group | 54/64 (84.4%) | 47/61 (77.0%) |
| S pyogenes | 21/23 (91.3%) | 16/18 (88.9%) |
| Pathogens | BAXDELA n/N |
Comparator n/N |
|---|---|---|
| Escherichia coli | 12/14 (85.7%) | 18/20 (90.0%) |
| Enterobacter cloacae | 12/14 (85.7%) | 10/11 (90.9%) |
| Klebsiella pneumoniae | 20/22 (90.9%) | 21/23 (91.3%) |
| Pseudomonas aeruginosa | 11/11 (100%) | 12/12 (100%) |
Melinta Therapeutics, Inc. - The Antibiotics Company
For additional information about BAXDELA, call Melinta Therapeutics at 1-844-MELINTA (1-844-635-4682) or contact the US Food and Drug Administration at 1-888-INFO-FDA (1-888-463-6332).
©2017 All rights reserved PP-BAX-US-0072 v1.1 Terms of Use